|
4/12/2023 0 Comments Complex ion bonding
Lewis bases have excess electron pairs which remain excess even after octet completion of the outermost electronic shell. CO 2, SO 2, NO 2, etc.Ī chemical species with the ability to donate an electron pair is called a Lewis base. Molecules with multiple bonds between two atoms of different electronegativity values e.g.Molecules with the capacity to absorb more than eight electrons e.g. 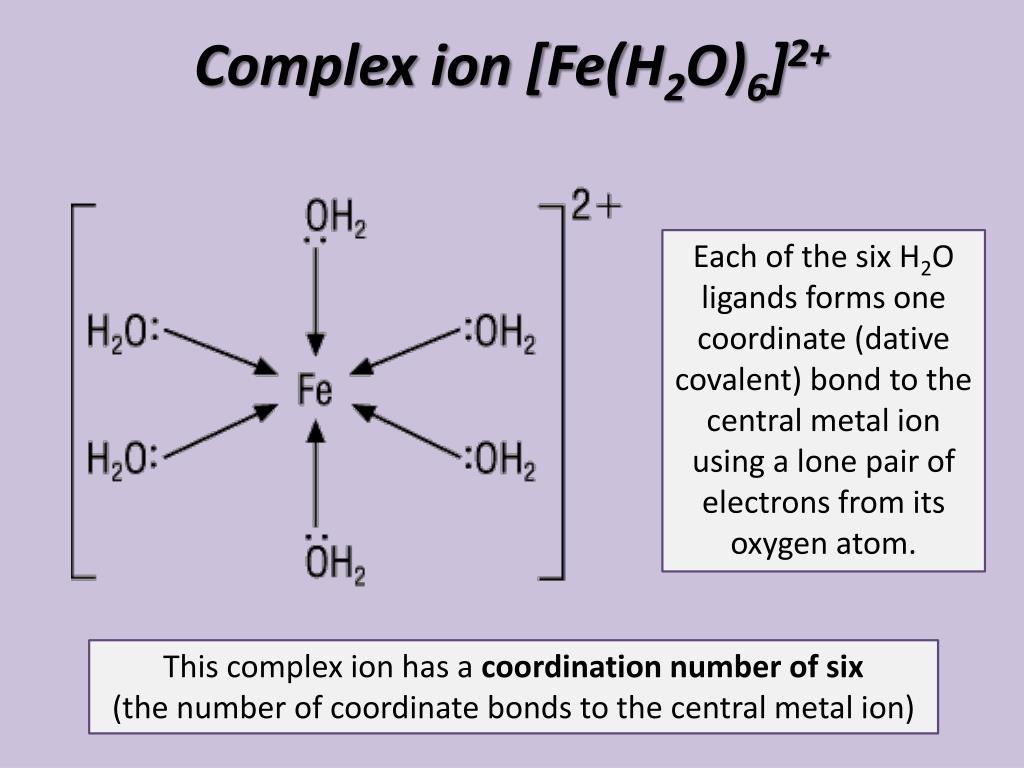
Atoms, ions, and molecules with incomplete octet e.g.All cations are lewis acids as they have the capacity to accept electron pairs. Basically, these species have empty orbitals in these elements which make them good electron acceptors. So, the electron-deficient species make a bond rather than staying alone.Ī chemical species with the ability to accept an electronic pair is called a Lewis acid. Although this disturbs the octet completion of that element, what better could come out of a lone electronic pair than the formation of a bond. The electron-donating species is always with an extra pair of electrons that can be donated. It is a covalent bond in which the sharing of electrons to make a bond is only observed by one of the bonded atoms. This bond pair is known as a covalent bond.Ī methane molecule is made by the mutual sharing of electrons between the carbon atom and hydrogen atoms. In 1916, Gilbert Newton Lewis (1875-1946) published his work on chemical bonds saying that a chemical bond is an electronic pair shared by two constituent atoms.Įach atom usually gives one electron in a bond pair. Usually, it happens in molecules where metallic ions are bonded to ligands. The coordinate covalent bond, also known as the dative bond, is a type of covalent bond where both electrons are from the same atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
